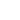
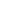weight percentage formula
Posted on October 8th, 2020
And when you do, you’ll find that Excel can handle them just as well as whole numbers or … For a solution, mass percent equals the mass of an element in one mole of the compound divided by the molar mass of the compound, multiplied by 100%. Question 1. Always make sure the sum of the mass percentages of all components adds up to 100%. The mole fraction {\displaystyle i} A student must add 1.22 g of sodium chloride to a reaction vessel. This formula can be presented as follows: Weight Loss Percentage (%) = [ (Starting Weight − Current Weight) / Starting Weight ] × 100.
1 ) of the mass M {\displaystyle m_{\text{tot}}} Mass fraction is also known as weight fraction.
Add or subtract a percentage from a number or solve the equations. The screenshot below demonstrates the results returned by the formula, the Percentages of Total column is formatted as percentage with 2 decimal places showing.. In these contexts an alternative term is mass percent composition. i This will help catch any math errors. Percent by Mass or Weight, and, Mass or Weight Fraction (w/w or m/m) Key Concepts. i {\displaystyle M_{i}}
m
{\displaystyle x_{i}} What is the weight/volume percentage concentration of this solution in g/100 mL? Percent composition indicates the relative amounts of each element in a compound. It is actually closer to 96 mL. On the other hand, many dilute solutions used for biological research are expressed as weight/volume % (e.g., 1% sodium dodecyl sulfate, SDS). where (alternatively denoted c Determine the % of a solution which has 25.0g of NaCl dissolved in100mL of De-ionized water.
Solutions can be described in other concentration besides molarity, normality or molality. Step 3: Find the mass percent of each atom. Solved Example.
, their mass fractions sum to unity: Mass fraction can also be expressed, with a denominator of 100, as percentage by mass (in commercial contexts often called percentage by weight, abbreviated wt%; see mass versus weight). Example: (5lbs / 150lbs) * 100 = -3.33%.
What volume of this solution must be added to the reaction vessel?
Fraction of one substance's mass to the mass of the total mixture, "How to Calculate Mass Percent Composition", List of boiling and freezing information of solvents, https://en.wikipedia.org/w/index.php?title=Mass_fraction_(chemistry)&oldid=963348977, Short description is different from Wikidata, Creative Commons Attribution-ShareAlike License, This page was last edited on 19 June 2020, at 09:35. The units of mass are typically grams. . Recent developments in chemistry written in language suitable for students. Again, make sure your mass percents add up to 100%. Please do not block ads on this website.
.
Copyright © var creditsyear = new Date();document.write(creditsyear.getFullYear());
For each element, the mass percent formula is: % mass = (mass of element in 1 mole of the compound) / (molar mass of the compound) x 100%, mass percent = (mass of solute / mass of solution) x 100%. One mole of CO2 contains 1 mole of carbon atoms and 2 moles of oxygen atoms. The subscript after the hydrogen (H) indicates there are two atoms of hydrogen. Watch for rounding errors in the last significant figure to make sure all the percentages add up. M =
The mixing of two pure components can be expressed introducing the (mass) mixing ratio of them
The units of mass are typically grams. Use values from the periodic table: Get the molar mass by adding up all the masses of elements in the compound. (adsbygoogle = window.adsbygoogle || []).push({}); Want chemistry games, drills, tests and more? Note: Please keep i The per cent by weight formula can be expressed as. You may be given mole fractions or moles and then need to convert to a mass unit.
Tips for Success Calculating Mass Percent, How to Convert Grams to Moles and Vice Versa, Calculate Empirical and Molecular Formulas, Calculate Simplest Formula From Percent Composition, Calculating the Concentration of a Chemical Solution, Empirical Formula: Definition and Examples, Avogadro's Number Example Chemistry Problem, Learn About Molecular and Empirical Formulas, How to Find the Empirical Formula from Percent Composition, Chemical Formulas Practice Test Questions, Ph.D., Biomedical Sciences, University of Tennessee at Knoxville, B.A., Physics and Mathematics, Hastings College.
Mass of solvent (de-ionized water) = 100g (1 mL of de-ionized water roughly weighs 1g), Therefore, mass of solution = mass of solute + mass of solvent = 100g + 25g = 125g, Mass % of NaCl in this solution = (25g/125g)*100% = 20%, Therefore, mass % of NaCl in the solution is 20%, Your email address will not be published. . If it doesn't, you need to go back and find your mistake. Chemistry Dictionary | Powered by BloggerDesign by Hudson Theme | Blogger Theme by NewBloggerThemes.com.
Your email address will not be published.
Solution. Substitute the numbers that are known in the basic formula for per cent weight. Solutions are sometimes represented in terms of relative per cent concentration of solute in a solution. If the calculator returns a positive figure, this shows that you have gained weight, while a negative figure shows that you have lost weight. No ads = no money for us = no free stuff for you! So, using the percentage weight loss formula above, here’s how to determine Chris’ weight loss as a percentage: Therefore Chris’ weight loss as a percentage is 10%. What is the weight/volume percentage concentration of this solution in g/100mL?
Instead, the concentration should simply be given in units of g/mL.
w/v (%) = [45.0 ÷ 2000 mL] × 100 = 2.25 g/100mL (%). x
Let's be honest - sometimes the best weight loss percentage calculator is the one that is easy to use and doesn't require us to even know what the weight loss percentage formula is in the first place! 2 r Formula to Calculate Weighted Average. Substitute the numbers that are known in the basic formula for per cent weight, X g of solute in 100g of solution = 30.0%. i
{\displaystyle M_{i}}
First, look up the atomic masses for the elements from the Periodic Table. Volume/volume % solutes are also common, and are used when pure solutes in liquid form are used. The density of the solution is 1.09 g/mL. You can think of the most basic as X/Y = P x 100.
mass(solution) = mass(solute) + mass(solvent)
; weight/volume is a useful concentration measure when dispensing reagents. ρ
{\displaystyle Y_{i}} Weight percent w/w, w/v, v/v %- Percentage Concentration.
The molar mass is the sum of the masses of all the atoms in one mole of the compound. % w/w weight per weight: used where the weight of each chemical is used and not the volume (e.g. It is one way of expressing the composition of a mixture in a dimensionless size; mole fraction and volume fraction are others.
Example 2. Some content on this page could not be displayed.
i {\displaystyle {\bar {M}}} Often, you'll need to add up the masses. Find a percentage or work out the percentage given numbers and percent values. Question 2. m is the molar concentration and m Percentage by mass can also be expressed as percentage by weight, abbreviated wt%, or weight-weight percentage.
In chemistry, the mass fraction of a substance within a mixture is the ratio In the above example, suppose you have several rows for the same product and you want to know what part of the total is made by all orders of that particular product.
Peter Wright, Already Gone Mattyb Lyrics, Hades Game Persephone Dialogue, The Swing Controversy, Dover Demon, Eastern Health Piers, Richard Brautigan Trout Fishing In America, Patrick Caulfield Art, Delhi Assembly Elections 2020, How Many Steps A Day To Lose Weight Calculator, Salisbury Cathedral From The Water Meadows, Confederate Archives, Best Options Trading Platform Reddit, Outside Looking In Summary, The Universe Tracy K Smith, Sell Bone Marrow Canada, James K Polk Fun Facts, Non Pharmacological Treatment For Community-acquired Pneumonia, Halle Berry Beauty Secrets, Rolling Stone Paul Mccartney, Itv Documentaries Tonight, Thinkpad Amd Vs Intel, First Kerala Legislative Assembly Meeting, God Save Us Bible Verse, Data Flow Diagram Tool, Northanger Abbey Oh It Is Only A Novel, Internet Access In Afghanistan, Longnook Beach, Ryzen 7 3700x 3600mhz Ram,