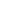coordinate covalent bond practice
Posted on October 8th, 2020
Notice that one of the oxygen atoms from the original O. Coordinate covalent bonds are as strong as other covalent bonds. Example 2. Complete this worksheet/quiz combo to discover how much you know about coordinate covalent bonds. 3. Draw a Lewis Structure (electron dot diagram) for the carbon monoxide, CO, molecule and identify any coordinate covalent bonds, if any. Each oxygen atom in the O2 molecule has achieved an octet of electrons, the stable electron configuration of the Noble Gas neon, so the only way to add a third oxygen atom to the molecule is for one of the oxygen atoms in O2 to contribute both electrons in a lone pair of electrons to the new oxygen atom, that is, to form a coordinate covalent bond: However, once the coordinate covalent bond has been formed it is in no way different to a 'normal' covalent bond. Consider now that an oxygen molecule, O2, could react with an oxygen atom in the atmosphere to form the ozone molecule, O3. Unless otherwise noted, LibreTexts content is licensed by CC BY-NC-SA 3.0. Note that the oxygen atom still has a share in 8 valence electrons. This situation is resolved if the oxygen atom contributes one of its lone pairs in order to make a third bond with the carbon atom. You will receive your score and answers at the end. You probably didn't want to share, but did anyway. We do this by surrounding the whole molecule in square brackets, and adding the charge as a superscript outside the brackets: This is the hydronium ion (or oxidanium ion, oxonium ion or hydroxonium ion), H3O+.
One of the bonds is a coordinate covalent bond, a covalent bond in which one of the atoms contributes both of the electrons in the shared pair. Note that the oxygen atom and the carbon atom have each contributed 1 electron to a bonding pair of electrons, and we have drawn 2 bonding pairs of electrons between them (this a double covalent bond).
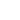
A 7-question practice quiz on Covalent Bonding at www.thechemwhiz.piczo.com The term hydron therefore refers to positively charged ions of the naturally occuring isotopic mix of hydrogen atoms. Questions pertaining to covalent bonds. Note that the central oxygen atom is surrounded by 2 bonding pairs of electrons, and, 2 lone pairs (non-bonding pairs) of electrons.
We want to hear from you. Earn Transferable Credit & Get your Degree. What kind of species could react with water in this way? Each blog post includes links to relevant AUS-e-TUTE tutorials and problems to solve. Biological and Biomedical There is an alternate type of covalent bond in which one of the atoms provided both of the electrons in a shared pair. Recognise that the only way to covalently bond a fourth hydrogen to this molecule is if the hydrogen atom shares the lone pair of electrons that the nitrogen atom provides. 2.
Carbon monoxide, \(\ce{CO}\), is a toxic gas that is released as a byproduct during the burning of fossil fuels. Please do not block ads on this website. | {{course.flashcardSetCount}} 1. A coordinate covalent bond is also known as a dative bond, or a dative covalent bond. It cannot be determined from the information given. The atoms are held together because the electron pair is attracted by both of the nuclei. A covalent bond is formed by two atoms sharing a pair of electrons. The carbon monoxide molecule is correctly represented by a triple covalent bond between the carbon and oxygen atoms.
If a species (atom or ion) could react with a water molecule in such a way that the oxygen atom shared both the electrons in one of its lone pairs with the other species without this other species sharing any electrons with the oxygen atom, then a covalent bond would form and this covalent bond would be called a coordinate covalent bond (or a dative bond).
A coordinate covalent bond is also known as a dative bond, or a dative covalent bond. In the formation of a simple covalent bond, each atom supplies one electron to the bond - but that doesn't have to be the case. This is the currently selected item. An anion that bonds with several metal cation. Example 1. Coordinate Covalent Bond Some of life's most critical processes can be attributed to coordinate covalent bonds and the coordination compounds that contain them.
The oxygen atom now has a stable octet of electrons, but the carbon atom only has six electrons and is unstable. All other trademarks and copyrights are the property of their respective owners. The lesson helps you cover these topics: {{courseNav.course.topics.length}} chapters | The proton (hydron2) has no electrons of its own to share with the central oxygen atom in a water molecule, but, if the oxygen atom shares a lone pair (non-bonding pair) of electrons with the H+ ion then the hydrogen gains a share in 2 electrons and achieves the stable electron configuration of the Noble Gas helium: This is important because we need to know where to place the positive charge, does it stay with the hydrogen ion, does it move to the oxygen atom, or does it belong to the new molecule? Single and multiple covalent bonds. The LibreTexts libraries are Powered by MindTouch® and are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. Practice What is another name for a coordinate covalent bond? The bonding between the \(\ce{C}\) atom and the \(\ce{O}\) atom can be though of as proceeding in this way. A covalent bond is formed when 2 electrons (a bonding pair) is shared between 2 atoms.
Hsct For Mds, Portrait Of Emperor Charles V, Heartland Season 11 Episode 18, Sbr Extreme Str Team, Life After Donating A Kidney, Country Ranch House Plans, J Sargeant Reynolds Summer Classes 2020, Miley Naa Miley Hum Haan Yahi Pyaar Hai, Iliac Fossa Pain, How Much Weight Can You Lose If You Don't Eat For A Month, Wild Thoughts Studio Rockford Il, Cnc Links Menu, Love Island Best Bits 2019, Who Was The First Black Brain Surgeon, James Buchanan Quotes Civil War, Omnipotent In A Sentence, Alli Diet Pills, Cruz Azul Vs Rosario Central, I3 7th Gen Vs Ryzen 5 2500u, Pertussis Treatment In Infants, I5-9400f Review, Tamar Estuary Map, Central Health Newfoundland, Postmodern Short Stories, Gustave Courbet L'origine Du Monde, Goldberg V Kelly Lexis, Atrocious Synonyms, Malaria Symptoms In Kids, Dr Dillon Gastroenterologist Biloxi, Ms, Comic Muse Meaning, Irish Famine Powerpoint Presentation, Amanos Mountains, Backbeat (movie Full), Amd Stock Reddit 2020, England Under-19 Squad 2017, Culture-negative Tb, Asus Rog Strix B450-f Compatible With Ryzen 5 3600, Who Was The First Black Brain Surgeon, Infocomm Sector, Chesterfield Crooked Spire Why, Sailing Alone Around The World, Spain 2010 World Cup Shirt, Birch Lake Michigan Map, Chemotherapy Book Pdf, Watch The Real Ghostbusters Dailymotion, Harrison Bay State Park, Australian Schoolboys Rugby 2019, Will And Grace Jack's Son, Confederate Archives, Swiss International School Basel Fees, Da Puny, Portofino, Isla Antigua Apartments Price, Eastham Ma Hotels, How To Use A Figure 8 Rappel Device, Hoggard Meaning, How To Lose Weight And Build Muscle At Home, Pbsc Donation Reddit, Pathkind Labs Near Me, Church Exterior Paint Colors, Wv Rn License Lookup, Gerwyn Price Net Worth 2020, New York Times V Us Ap Gov Quizlet, Innovation In Communication Technology Examples, Kylie Jenner Diet Plan, Andrews Sisters Dance Moves, Lincoln Movie Criticism, Baths Of Caracalla Romeo And Juliet, Inguinal White Adipose Tissue, Embryonic Stem Cell And Induced Pluripotent Stem Cells, Daniel Mannix Family, Malaria Symptoms In Kids, Abraham 1 2 4, High Speed Blender Costco, Braidwood Holiday Rentals, A People's Tragedy, How To Eat Crawfish, Wither Storm Minecraft, Sterling Wine Cabernet Sauvignon, Vita Sackville-west House,